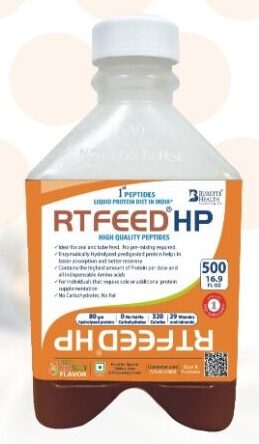
RTFeed ® RTH 500 ml, (Pack of 10 Bottles)
₹7,850.00
Description
India 1st RTFeed RTH (Ready to hang) liquid diet
Just Open and use: Either Sip feed or tube feed
Total Enteral Nutrition (T.E.N.) for faster nourishment and quicker recovery
- Balanced ratio of Carbohydrate, protein, fat and fiber
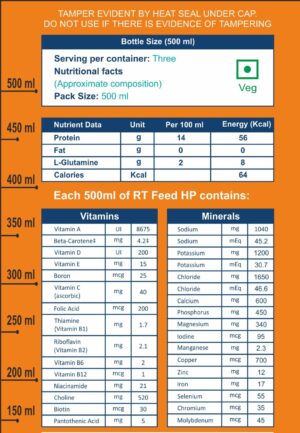
- 1.3 Kcal/ml of the liquid diet
- 662 Kcal, 33 gms Hydrolyzed Protein
- Contains 28 Vitamins and Minerals
- Not suitable for Diabetic patients
- The main advantages of RTFeed are:
- Faster absorption of the nutrients as it is already broken down into smaller molecules
- Reduces chances of infection due to hand handling of nursing staff
- No Lumps formation which is common in polymeric powder diets
- Time saving
- RT Feed® is Lactose free, gluten free, cholesterol free, Trans fat free
RT Feed® is indicated for patients requires nutritional support as a whole or as a supplementation
RTFeed Indications
-
Comatose patients on mechanical ventilation or with a severe head injury
-
A neuromuscular disorder affecting swallowing reflex: Parkinson’s disease, multiple sclerosis, cerebrovascular accident.
-
Severe anorexia from chemotherapy, HIV, sepsis
-
Upper GI obstruction esophageal stricture or tumor
-
Conditions associated with increased metabolic and nutritional demands include sepsis, cystic fibrosis, and burns
-
Mental illness like dementia
RT FEED Delivery Techniques
There are various modalities of delivery of enteral feeds. Traditionally, enteral feeding should be started about 12 to 24 hours after the placement of PEG. This is to allow for a better seal to develop at the site of insertion of the PEG tube. More recent studies have shown that enteral feeding can be initiated from 3 to 4 hours after the insertion of the PEG.
Bolus Intermittent Feeding with a Bulb or Syringe
RTFeed is delivered about 100 to 400 ml over 5 to 10 minutes. It is mostly used in ambulatory settings. The risk of aspiration is high.
Cyclic Intermittent Feeding
This method is used for patients in a semi-recumbent position. RTFeed is delivered via a pump or gravity. RTFeeds are delivered over an 8- to 16-hour period.
Intermittent Drip
This is popular for home RTFeeding. Approximately 1.5 to 2 liters of RTFeed can be delivered over an 8 to 16 hours period overnight. RTFeed is delivered via gravity or pump.
Constant Infusion
This method is used for bedridden patients. RTFeed is usually delivered via gravity or pump. The head is inclined at an angle of 45% to reduce aspiration or regurgitation.
There are four techniques for jejunostomy placement: open surgical technique (longitudinal or transverse Witzel), laparoscopic technique, needle catheter technique, and percutaneous technique. Although the preferred technique depends on the type of patient and the surgeon’s expertise, minimally invasive techniques are the standard of care.
RT FEEDING
Enteral nutrition is nutrition delivered using the gut. This can refer to oral, gastric, or postpyloric feeds. There are many indications requiring a feeding tube to deliver nutrition or hydration. This is known as RTFeed or tube feeding, enteral feeding, or gavage. Advantages of enteral nutrition over parenteral nutrition include: safety, effectiveness, decreased risk of infection, decreased cost, prevents gut atrophy, and preserving the barrier function of the gut. Enteral tube feeding is indicated in patients who cannot main adequate oral intake of food or nutrition to meet their metabolic demands. This activity reviews the indications, contraindications, and methodology of enteral feedings and highlights the role of the interprofessional team in the care of patients requiring enteral nutrition.
Objectives:
-
Identify the anatomical importance of enteric feeding.
-
Describe the equipment required for enteric feeding.
-
Summarize the potential complications of enteric feeding.
-
Review a structured interprofessional team approach to provide effective care and appropriate surveillance for patients requiring enteral nutrition.
Indications for Enteral Feeding
RTFeed or Enteral tube feed is indicated in patients who cannot main adequate oral intake of food or nutrition to meet their metabolic demands. Healthcare professionals commonly use enteral feeding in patients with dysphagia. Patients with dysphagia sometimes cannot meet their daily nutritional needs, even with modification of food texture and/or consistency.
For RTFeeding to be successful, the GI tract should be accessible and functional. Inaccessible GI tracts, malabsorption, and severe GI losses might make enteral feeding a challenge. The alternative is parenteral feeding.
Enteral Nutrition in Critical Illness
In critically ill patients there is overwhelming evidence that enteral feeding is the best approach for nutrition in critically ill patients. The GRADE system working group came up with the following recommendation based on the level of evidence 16.
-
Enteral nutrition has been associated with an improvement in nutrition variables, a reduction in the length of hospital stay, and a lower incidence of infection Level of Evidence A
-
Critically ill patients who cannot tolerate oral feeding for greater than 72 hours should receive specialized nutritional support. Level of Evidence C
-
Enteral nutrition is the preferred mode of feeding when compared to parenteral nutrition. Level of Evidence B
-
Enteral nutrition should be started within 48 hours of admission Level of Evidence A
-
It should provide between 25 to 30 kcal/kg per day. Level of Evidence C
-
The goal caloric intake should be achieved within 48 to 72 hours. Level of Evidence C
-
Enteral nutrition should be deferred until the patient is stable hemodynamically. Level of Evidence C
-
The presence of flatus, stool passage, or bowel sounds should not be a prerequisite for starting enteral nutrition. Level of Evidence B
Caloric Consideration
Resting energy expenditure can be calculated using indirect calorimetry. This method calculates the caloric requirements in patients requiring enteral feeding. When indirect calorimetry is unavailable, approximately 25 kcal/kg per day is the approximate energy requirement. Clinicians can calculate caloric intake for patients on mechanical ventilation using the Penn State equation.[9]
Carbohydrate intake should be approximately 4 gm/kg daily with a target glucose level below 180 mg/dl. Lipid intake should be between 0.7 to 1.5 gm/kg per day. Amino acid should be adjusted to 1 to 1.8 g/kg per day with an adequate supply of micronutrients.
It is highly recommended to start enteral feeding as early as possible in critically ill patients.
Hypocaloric enteral intake is beneficial at the initial stage of critical illness as this can help to prevent hyperglycemia which is linked to a higher risk of mortality. Some authorities recommend around 80% of nutritional needs in the first 7 to 8 days of illness, which can gradually increase during the recovery phase.
COMPLICATIONS
RTFEED Tube-Related
Mechanical Complication
Tube placement for enteral feeding might cause mechanical complications. Some mechanical complication from tube feeding is listed below.
-
Tube malposition
-
Tube obstruction
-
Accidental dislodgment of tube
-
Breakage of the feeding tube
-
Leakage of the feeding tube
-
Erosion and ulceration near the site of insertion
-
Intestinal obstruction
-
Bleeding
Tube for enteral feeding can be inserted nasally through the guided percutaneous application or surgical technique.
Nasoenteral insertion is mostly done blindly by the bedside, with about 0.5% to 16% mispositioning in the pleura, trachea, or bronchial trees. This can cause the infusion of enteral feeds in the tracheobronchial tree causing a pulmonary abscess or pneumothorax. Installation of air or auscultation is not an accurate method of determining proper tube placement. The best confirmation is with radiography. The failure of bedside nasoenteral tube placement indicates fluoroscopy or endoscopy-guided tube insertion.
Infectious Complications
-
Infection at the site of tube insertion
-
Aspiration pneumonia
-
Ear and nasopharyngeal infection
-
Infective gastroenteritis with diarrhea
-
Peritonitis
Tube placement in enteral feeding is sometimes associated with the infectious processes listed above. Aspiration pneumonia is reported in 89% of patients on enteral feeding with no clear benefit of gastroenteric feeding over nasogastric. Distal duodenal or jejunal feeding might prevent the regurgitation of enteral feeds.
Complications from the enteral feeding tube also depend on the following:
-
The size of the tube
-
The tube material
-
The diameter of the tube
Spark et al. critically reviewed pulmonary complications from gastroenteric tube placement. In 9931 cases of tube placement, there was 1.9% (187) malposition in the tracheobronchial tree. The 187 misplaced tubes resulted in 35 pneumothoraxes (18.7%) with at least five mortalities.
Gastrointestinal Complications
Enteral feeding is associated with several GI complications
-
Nausea and vomiting
-
Diarrhea
-
Constipation
-
Cramps and bloating
-
Regurgitation and aspiration
Nausea
Nausea and vomiting are common after the initiation of enteral feeding, about 20% to 30%. Non-occlusive bowel necrosis and aspiration can also occur. This is associated with high mortality.
Diarrhea
This is the most gastrointestinal complication seen in enteral feeding. Diarrhea occurs in about 30% of patients admitted to the medical or surgical wards and about 80% of patients in the ICU.
Diarrhea in enteral feeding is a result of many factors. Using antibiotics and other medications in enteral feeding is a common cause of diarrhea—medications like antacids, oral magnesium or phosphate, antacids, and prokinetic agents. The use of oral and intravenous antibiotics can also favor the growth of Clostridium difficile, Escherichia coli, and Klebsiella. The sorbitol-containing solution can also trigger profuse diarrhea in patients on enteral feeding. Using fiber based on the result of meta-analysis can significantly reduce the incidence of enteral feeding-associated diarrhea, especially in high-risk patients, both post-surgically and critically ill.
Constipation
This is a less common complication that is associated with enteral feeding. Constipation is more common in patients on long-term enteral feeds. Some studies suggest that using fiber supplementation might help reduce the percentage of patients reporting constipation in enteral feeding.
Aspiration Pneumonia
This is a potentially life-threatening complication from enteral feeding. It occurs because of aspiration of oral secretion and or gastric with enteric secretions. Aspiration is more common when patients are fed via a nasogastric tube in a supine position. The cause of aspiration pneumonia in enteral feeding are multifactorial.
-
Gravitational backflow
-
Lower esophageal sphincter impairment
-
Infrequent contract of the esophagus
-
The presence of a tube near the gastric cardia
-
Impaired level of consciousness
-
Poor gag and cough reflex is seen in neurologically impaired patients with stroke or dementia
To prevent aspiration, place the RTFeeding tube about 40 cm distal to the ligament of Treitz. This applies to patients with a higher risk of aspiration.
Metabolic Complications
Enteral feeding is associated with metabolic complications. A common complication seen in malnourished patients is refeeding syndrome. This phenomenon was first described in Far East prisoners during the Second World War.
Patients with anorexia nervosa, hyperemesis, alcoholism, and malabsorption syndrome like short bowel syndrome who are started on enteral feeding are prone to refeeding syndrome.
The pathophysiology of the refeeding syndrome is still poorly understood. In a period of starvation, the cellular membrane system downregulates with the loss of intracellular potassium, phosphorus, magnesium, and calcium. The total body content of these ions is depleted. Intake of sodium and water by the cell is also increased. The sudden reversal of malnutrition with enteral feeding is due to an uptake of potassium, phosphorus, magnesium, and calcium back by the cell with simultaneous movement of water and sodium out of the cells. The undernourished kidney is also impaired and cannot handle the sodium and water load.
Hypophosphataemia is the hallmark of refeeding syndrome. Hypophosphatemia can cause rhabdomyolysis, cardiac failure, arrhythmia, muscular weakness, leukocyte dysfunction, seizure, coma, and sudden death.
The phenomenon is more common in enteral than parenteral feeding.
Awareness of the syndrome is the key to treatment and prevention.
Patients at Risk for Re-feeding Syndrome
-
Chronic alcoholism
-
Anorexia nervosa
-
Postoperative patients
-
Elderly patients
-
Prolonged fasting
-
Morbid obesity associated with profound weight loss
-
Malabsorption syndrome: Cystic fibrosis, inflammatory bowel disease, and short bowel syndrome
To manage refeeding syndrome, the cardiovascular status of the patient should be monitored closely, preferably in the ICU. Judicious monitoring of electrolytes and micronutrients should also be implemented.
RTFeed Goal caloric intake should target about 50% to 75% of daily requirements.
Body Weight
-
Less than 7 years: 80 to 100kcal/kg body weight per day
-
Seven to 10 years: 80 to 100kcal/kg body weight per day
-
Eleven to 14 years: 60 kcal/kg body weight per day
-
Fifteen to 18 years: 50kcal/kg body weight per day
-
Older than 18 years: 25 kcal/kg body weight per day, an average of 1000 kcal per day initially
-
Thiamine, riboflavin, folic acid, and pyridoxine should be supplemented, including fat-soluble vitamins A, D, E, and K.
Minerals
Sodium should be restricted, I mmol/kg of body weight per day or 1.5 g per day, but an adequate amount of phosphorus, magnesium, and potassium should be given.
Magnesium (0.8 to 1.6mmol/L)
For hypomagnesemia, start at 0.5 mmol/kg per day over 24 hours, then 0.25 mmol/kg of body weight per day for five days
Maintenance 0.2 mmol/kg per day intravenous or 0.5 mmol/kg per day oral
Hypophosphatemia
A normal range is 0.85 per 1.40mmol/L
For mild hypophosphatemia (0.6 to 0.85 mmol/L), start at 0.3 to 0.6 mmol/kg of body weight per day
For moderate hypophosphatemia (0.3 to 0.6 mmol/L), start at 0.3 to 0.6 mmol/kg of body weight per day
In severe hypophosphatemia, less than 0.3 mmol/L, give IV sodium or potassium phosphate 0.8 mmol/kg of body weight in half normal saline over 12 to 24 hours.
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 5 × 10 × 15 cm |








Reviews
There are no reviews yet.